FAQ Medikamente Info Austria
2. In which languages is the "Medikamente Info Austria" available?
“Medikamente Info Austria” can be used in German and English.
3. How often is the data in the "Medikamente Info Austria" updated?
The data is updated once a day. The time of the last update can be seen in the “Medikamente Info Austria” in the search area.
4. Can I search using wildcards?
Yes, you can use * to optimise your search in text fields.
If *
- is specified before the search value, hits are returned in which the search value appears somewhere in the name.
- specified after the search value, results are returned that begin with the search value.
- if * is used more than once, results can be restricted even more specifically and both search options can be combined.
5. General information on the Medicinal product index
Legal basis: Ordinance of the Federal Minister of Health on the Austrian Medicinal Product Index 2013.
The details of all medicinal products authorised/registered in Austria can be searched using the “Medikamente Info Austria”.
You can search for several terms and thus determine the scope of the results data.
For medicinal products that have been authorised centrally - recognisable by the authorisation number beginning with EU/.. - the corresponding information can be found on the website of the EMA (European Medicines Agency).
7. What data is displayed in the hit list of the Medicinal product index?
- Name: Name of the medicinal product in accordance with Section 1(4)(14) of the Austrian Medicinal Products Act (AMG).
- Active substance(s): Medicinally active ingredients according to Section 1 paragraph 4 number 4a of the Austrian Medicinal Products Act (AMG).
- Package leaflet: Currently valid Package leaflet in accordance with Section 16 of the Austrian Medicinal Products Act (AMG) (only displayed for national marketing authorisations and can be viewed/downloaded).
- Holder: Name of the holder a marketing authorisation/registration or authorisation of the parallel import, in accordance with Section 2 (13a) of the Austrian Medicinal Products Act (AMG).
- Prescription required: Indication of whether a medicine is prescription-only or not (more information on prescription status can be found in the detailed view of the selected medicine).
8. What data are displayed in the "Details" tab of the detailed view of the Medicinal products index?
- Name: Name of the medicinal speciality in accordance with Section 1 paragraph 4 number 14 of the Austrian Medicinal Products Act (AMG).
- Active substance(s): Medicinally active ingredients in accordance with Section 1 paragraph 4 number 4a of the Austrian Medicinal Products Act (AMG). If a medicinal product contains several active ingredients, these are separated by the separator ‘I’.
- ATC code: Anatomical Therapeutic Chemical Classification according to WHO (World Health Organization).
- Prescription status: The prescription status determines whether a medicine must be prescribed by a doctor or whether it can be purchased from a pharmacy without a doctor's prescription.
Legal basis in the currently valid version: Medicinal Products Act (AMG), Compulsory Prescription Act, Compulsory Prescription Regulation, Narcotic Drug Act, Narcotic drugs decree, Psychotropic substances decree. - Supply status: Supply regulations according to Section 57 of the Austrian Medicinal Products Act (AMG).
- Narcotic drug: Labelled with Yes if at least one active substance is subject to the Narcotic Drug Act.
- Psychotropic: Labelled with Yes if at least one active substance is subject to the Psychotropic substances decree.
- Holder: Name of the holder a marketing authorisation/registration or authorisation of the parallel import, in accordance with Section 2 paragraph 13a of the Austrian Medicinal Products Act (AMG).
- Pharmaceutical dose form: Pharmaceutical form of a medicinal product.
- Strength: Strength of a medicinal product.
- Marketing cessation notified: Labelled with Yes if the BASG has been notified of a temporary or permanent cessation of placing on the market in accordance with Section 21 paragraph 2 of the Austrian Medicinal Products Act (AMG).
- MR/DC/CP number: Product-related number consisting of the country code of the Reference Member State, the domain (human or veterinary), the product number and the strength number, e.g. AT/H/1234/001. In the case of centralised marketing authorisations, the EMA ‘core number’ is displayed, e.g. EMEA/H/C/xxxxxx.
- MA number: Unique number for the medicinal product that is assigned upon authorisation and remains identical throughout its life. Authorisation numbers beginning with EU indicate centrally authorised medicinal products.
- Authorisation date: Date on which the authorisation/registration was granted.
- Drug category: Category of a medicinal product.
- Vaccine: Labelled with Yes if the medicinal product is a vaccine.
- Allergen: Labelled with Yes if the medicinal product is an allergen product.
- Additional monitoring: The European Union has introduced labelling for medicinal products that are additionally - i.e. particularly closely - monitored. These medicinal products are labelled in the Information for healthcare professionals and in the package leaflet (formerly package insert) with a black triangle ▼ standing on its tip. See also https://www.basg.gv.at/en/companies/marketing-authorisation-life-cycle/faq-marketing-authorisation-life-cycle/black-triangle
- Mandatory for batch release?: Restriction for the batch release obligation in accordance with Section 26 paragraph 1 of the Austrian Medicinal Products Act (AMG). see also www.basg.gv.at/en/companies/marketing-authorisation-life-cycle/faq-marketing-authorisation-life-cycle/batch-release.
- Exception for batch release?: Restriction for an exemption from batch release in accordance with Section 26 paragraph 4 of the Austrian Medicinal Products Act (AMG). see also https://www.basg.gv.at/en/companies/marketing-authorisation-life-cycle/faq-marketing-authorisation-life-cycle/batch-release.
- Authorised in Liechtenstein?: Medicinal product authorised in accordance with BGBl. III Nr. 126/2010 (Agreement between the Austrian Federal Government and the Government of the Principality of Liechtenstein concerning the automatic recognition of human and veterinary medicinal products authorised or registered in Austria in Liechtenstein).
- EU databases:
- Documents on centrally authorised proprietary medicinal products can be found on the website of the European Commission.
- Information on existing parallel distributions can be found at https://iris.ema.europa.eu/registerpd/.
- Further information on medicinal products that have been authorised under the mutual recognition procedure (MRP) or decentralised authorisation procedure (DCP) can be found in the Mutual Recognition Index (MRI). Enter the value from the MR/DC/CP number column in the ‘Search’ field at the top right, e.g. AT/H/1234/001. Attention: no EMA ‘core number’ allowed.
- Further information on veterinary medicinal products (veterinary medicinal specialities) can also be found on the website of the European Union. - Domain: Indication of whether the medicinal product has been authorised for humans or animals.
- Animal species: For veterinary medicinal products is indicated in each case:
- Route of administration
- Dosage
- Tissue
- Withdrawal Period: <waiting time> <unit>
- Comment
9. Which documents are displayed in the "Documents" tab in the detailed view of the Medicinal product index?
For national marketing authorisations, the following documents are listed and can be displayed/downloaded if they are available:
- Package leaflet (national translation)
- Summary of Product Characteristics: SmPC (national translation)
- National Public Assessment Report (NPAR)
- Risk Management Plan (RMP Summary)
According to Art 27 Austrian Medicinal Products Act AMG the Austrian Medicinal Product Index is published by BASG (Federal Office for Safety in Health Care) where defined data of all authorised and registered medicinal product including their product informations (SmPC and PL) can be found.
In addition, the national public assessment report (NPAR), which is derived from the assessment during the evaluation process, is made available to the public after deletion of commercially confidential information.
PARs of European evaluation processes (MR/DC or centralised) are published in English, those of national procedures in German.
- If Austria is Concerned Member State (CMS), a link to the Mutual Recognition Index is published. The maintenance of this publication is the responsibility of the Reference Member State (RMS).
- If the medicinal product is authorised in a centralised procedure, a link to the European Medicine Agency's website is published. The maintenance of this publication is the responsibility of the EMA.
10. Why are no instructions for use and Summary of Product Characteristics (SmPC) linked for centrally authorised products?
The valid Package leaflet (PL) and Summary of Product Characteristics (SmPC) are available in EMA databases.
The “Community Register” is stored as a hyperlink and links to the following URL: http://ec.europa.eu/health/documents/community-register/html
A corresponding search can be carried out there if the hyperlink does not lead directly to the product page.
The Package leaflet and Summary of Product Characteristics (SmPC) can be downloaded individually in chronological order in the ‘Annexes’ section.
11. Can results from the Medicinal product index be exported?
Instructions for the BASG Website – Opening CSV Files from the Arzneispezialitätenregister
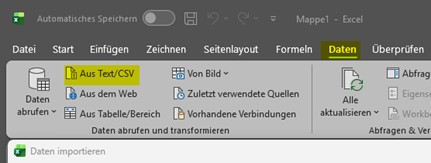
When downloading datasets from the Arzneispezialitätenregister, the information is provided exclusively in CSV format. Since CSV files do not contain formatting or layout information, opening them directly by double click may result in incorrect or irregular display within Microsoft Excel.
To ensure that the data is shown correctly and in a structured way, it is recommended to import the file using Excel’s data import function.
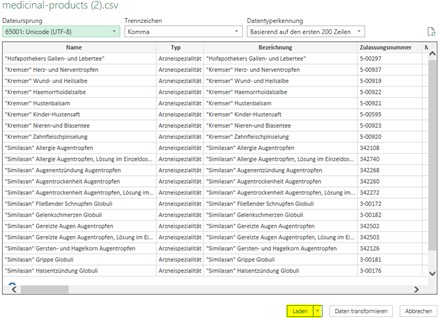
To do this, start Excel and select the “Data” tab in the upper menu. Using the “From Text/CSV” option, choose the CSV file previously downloaded. Excel will display a preview of the content and will automatically detect proper separators and formatting.
If display issues occur despite this procedure, they are often caused by differing system regional settings or by the fact that CSV files cannot always be interpreted correctly when opened directly.
Using the described import method is therefore the most reliable approach for correctly displaying and processing datasets exported from the Arzneispezialitätenregister.
12. Distribution restrictions register (general register)
All reported procedures are presented in the "Shortages index" - it is therefore the common register. Notifications concerning prescription-only medicines are checked by the BASG before publication.
Not included are notifications pursuant to Section 21 (2) of the Austrian Medicines Act as amended.
The notifications are automatically published in the "Shortages index" on the following day from the actual start of the shortage.
The reporters are responsible for the accuracy of the data. The BASG does not have information on existing stocks in the distribution chain (i.e. wholesalers, pharmacies).
14. What data is displayed in the hit list of the Shortages index?
- Name: Name of the medicinal speciality in accordance with Section 1 paragraph 4 number 14 of the Austrian Medicinal Products Act (AMG).
- Active substance(s): Medicinally active ingredients according to Section 1 paragraph 4 number 4a of the Austrian Medicinal Products Act (AMG).
- Status:
- Date of report.
- Applicant.
15. What data is displayed in the "Shortages index" tab in the detailed view?
- Active substance(s): Medicinally active ingredients in accordance with Section 1 paragraph 4 number 4a of the Austrian Medicinal Products Act (AMG).
- Strength: Strength of a medicinal product.
- Pharmaceutical dose form: Pharmaceutical form of a medicinal product.
- Domain: Indication of whether the medicinal product has been authorised for humans or animals.
- ATC code: Anatomical Therapeutic Chemical Classification according to WHO (World Health Organization).
- Reason
- Holder: Name of the holder of the marketing authorisation/registration or authorisation of the parallel import, according to Section 2 paragraph 13a of the Austrian Medicinal Products Act (AMG).
- Telephone number of holder
- MA number: Unique number for the medicinal product that is assigned upon authorisation and remains identical throughout its life. Authorisation numbers beginning with EU identify centrally authorised medicinal products.
- Important BASG-information
- Information letter to healthcare professionals
- Last modification date
- Date of report
- Legal basis of the notification
- Export ban
- Availability of packs
16. Can results from the Shortages index be exported?
Instructions for the BASG Website – Opening CSV Files from the Arzneispezialitätenregister
When downloading datasets from the Arzneispezialitätenregister, the information is provided exclusively in CSV format. Since CSV files do not contain formatting or layout information, opening them directly by double click may result in incorrect or irregular display within Microsoft Excel.
To ensure that the data is shown correctly and in a structured way, it is recommended to import the file using Excel’s data import function.
To do this, start Excel and select the “Data” tab in the upper menu. Using the “From Text/CSV” option, choose the CSV file previously downloaded. Excel will display a preview of the content and will automatically detect proper separators and formatting.
If display issues occur despite this procedure, they are often caused by differing system regional settings or by the fact that CSV files cannot always be interpreted correctly when opened directly.
Using the described import method is therefore the most reliable approach for correctly displaying and processing datasets exported from the Arzneispezialitätenregister.
17. What is meant by "Export ban index"?
The export ban index corresponds to the "Catalogue according to the regulation on ensuring the provision of medicinal products according to § 57a para. 2 Austrian Medicines Act". The catalogue is based on the same data as the "Shortages catalogue", but restricted to those medicinal products which have been banned from export to another contracting party of the European Economic Area.
18. What is meant by "Export ban"?
According to § 5 para. 1 of the regulation on ensuring the provision of medicinal products, export to another contracting party of the European Economic Area (EEA) is prohibited for reasons of public health protection. Excepted from this is the export for an individual named patient for the immediate prevention of a threat to life or serious damage to health ("Named Patient Use"). Third countries are exempt from the export ban. This is solely an export ban to the EEA, which means that authorised wholesaler may not trade those medicinal products from Austria to the EEA.
19. What is the data and legal basis for the "Export ban index"?
Legal basis in general: Regulation on ensuring the provision of medicinal products according to § 57a para. 2 Austrian Medicines Act.
Legal basis for the export ban index: Catalogue according to § 1 para. 3 Regulation on ensuring the provision of medicinal products, which must be published by BASG on its website for general access.
Data basis: After reviewing the notification, BASG must publish the affected medicinal product at the earliest on the day on which the shortage actually occurs.
20. What data is displayed in the BASG announcements?
BASG decisions and variations to medicinal products that may be relevant to users or pharmacists are published on a daily basis.
Further information can be found at https://www.basg.gv.at/en/about-us/basg-publications
21. What can be searched for and how can the list of results be narrowed down?
- Observation object (the name of a medicinal product) or marketing authorisation number (of a medicinal product)
- Period (of approval)
In the advanced search, additionally
- ATC code
- Reference product
- Holder
- Classification
- Procedure type
- Type
- Decision type
23. What data is displayed in the details?
- Name (corresponds to object): Name of the medicinal product in accordance with Section 1 (4) No. 14 of the Austrian Medicinal Products Act (AMG)
- Previous name: if the name has been changed, the previously valid name is displayed
- MA number: Unique number for the medicinal product that is assigned upon authorisation and remains identical throughout its life. Authorisation numbers beginning with EU indicate centrally authorised medicinal products.
- Procedure number: technically unique
- Procedure type: Type of application
- Procedure closed date: Approval date for application
- Classification: For applications for changes, the subject of the change is published in accordance with Commission Regulation (EC) No. 1234/2008 of November 24, 2008, on the examination of changes to the authorization of medicinal products for human and veterinary use, or Section 24 of the Austrian Medicinal Products Act (AMG).
- Holder: Name and address of the holder of the authorization/registration/approval of the parallel import, in accordance with Section 2 (13a) of the Austrian Medicinal Products Act (AMG).
- Previous holder: In the event of changes to the holder, the previously valid holder is displayed.
- ATC code: Anatomical Therapeutic Chemical Classification according to the WHO (World Health Organization)
- Prescription status: The prescription requirement is the determination of whether a medicinal product must be prescribed by a doctor or whether it can be purchased in a pharmacy without a doctor's prescription.
- Legal basis in the currently valid version: Austrian Medicinal Products Act (AMG), Compulsory Prescription Act, Compulsory Prescription Regulation, Narcotic Drug Act, Narcotic drugs decree, Psychotropic substances decree.
- Previous prescription status: If there are changes to the prescription requirement, the previously valid prescription status is displayed.
- Reference product: Reference drug in accordance with Section 1 (19) Austrian Medicinal Products Act (AMG) for generic drugs.
- Type: Decision or decision correction
- Decision type: Basis for the decision in accordance with Austrian Medicinal Products Act (AMG)/ Austrian Veterinary Medicinal Products Act (TAMG)
- Result: Result of the vote in the electronic approval process
- Approved on: Date of completion of the electronic approval process
- Negative decision: Rejection of the application
- Documents: Currently Package leaflet and Summary of Product Characteristics (SmPC) as a PDF file
24. What data is displayed in the BASG votes?
After a decision has been made, the results are listed on a daily basis in an electronic approval process.
Further information can be found at: https://www.basg.gv.at/en/about-us/basg-publications
27. What data is displayed in the details?
- Observation object: can be the name of a drug
- Procedure number: technically unique number of the application
- BASG number: technically unique number for the electronic approval process
- MA number: Unique number for the medicinal product that is assigned upon authorisation and remains identical throughout its life. Authorisation numbers beginning with EU indicate centrally authorised medicinal products.
- Holder/Applicant for clinical trials: Name and address of the holder of the authorisation/registration/approval for parallel import, in accordance with Section 2 (13a) of the Austrian Medicinal Products Act (AMG).
- Sponsor: for clinical trials
- Reference product: Reference medicinal product in accordance with Section 1 (19) Austrian Medicinal Products Act AMG for generic medicinal products
- Type: Decision, non-prohibition, decision (correction of decision), non-prohibition (correction of decision)
- Type of decision: Basis for the decision in accordance with Austrian Medicinal Products Act (AMG)/ Austrian Veterinary Medicinal Products Act (TAMG)
- Result: Result of the vote in the electronic approval process
- Approved on: Date of completion of the electronic approval process
- Negative decision: Rejection of the application